-

CIP
Systems
Solutions de nettoyage pour l'industrie alimentaire, pharmaceutique et cosmétique
La transformation des aliments ou, plus encore, la production de produits cosmétiques ou pharmaceutiques dans le respect de normes élevées de qualité et de sécurité ne sera pas possible sans des processus de nettoyage efficaces, reproductibles, enregistrables et durables. Cela ne signifie pas seulement l'élimination de la saleté visible, mais plutôt l'élimination des contaminations invisibles pour une production sûre et hygiénique. Les solutions du système CIP de ProXES conviennent aussi bien à l'industrie alimentaire qu'à l'industrie pharmaceutique et à l'industrie des produits de haute technologie. Notre succès en matière de nettoyage est basé sur TACT - la combinaison du temps, de l'action, de la chimie et de la température afin d'assurer la qualité du produit et la disponibilité de l'usine.
What is a CIP System?
A CIP system is an automated cleaning technology used to clean production equipment internally without disassembling the machinery. Cleaning liquids such as water, detergents, and sanitizing solutions circulate through tanks, pipelines, and processing units to remove residues and contaminants.
Compared to manual cleaning methods, clean in place CIP systems provide controlled and repeatable cleaning cycles that improve hygiene and operational efficiency. Automated monitoring of cleaning parameters also allows cleaning processes to be documented and validated.
CIP systems are widely used in industries such as:
• Food and beverage processing
• Pharmaceutical manufacturing
• Cosmetics and personal care production
• Dairy and beverage industries
By automating the cleaning process, a CIP cleaning system helps manufacturers maintain reliable hygiene standards and efficient production workflows.
Typical CIP Process Steps
Industrial CIP systems follow a defined cleaning sequence designed to remove product residues, microorganisms, and contaminants from process equipment. These automated cleaning cycles allow tanks, pipelines, and processing units to be cleaned without dismantling the installation.
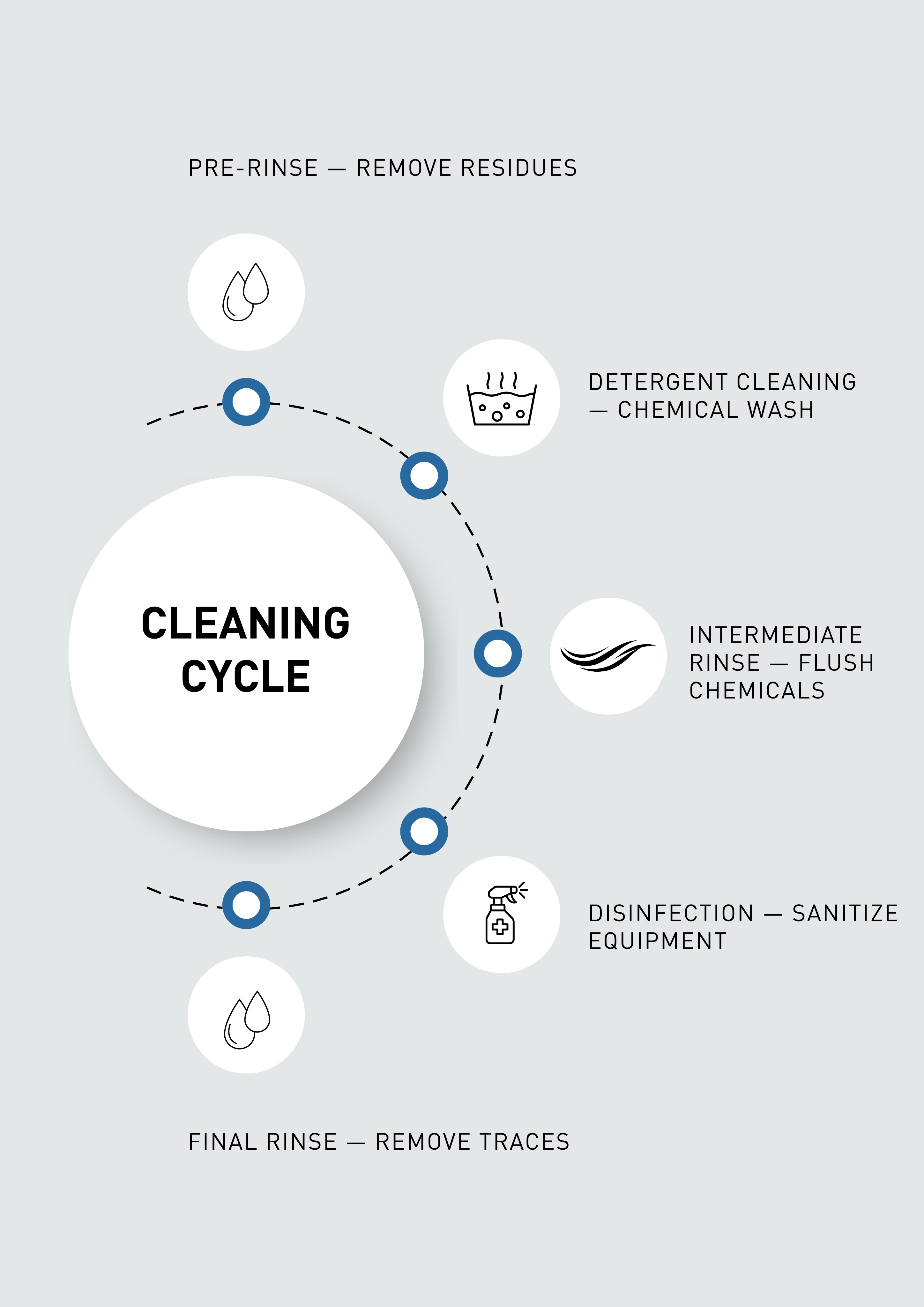
Cleaning Cycle Overview
A typical CIP cleaning cycle includes several stages:
• Pre-Rinse: Water removes loose product residues from the system.
• Detergent Cleaning: Alkaline or acidic cleaning agents dissolve fats, proteins, sugars, and other deposits.
• Intermediate Rinse: Remaining chemicals and dissolved residues are flushed out.
• Disinfection / Sanitization: Sanitizing agents eliminate microorganisms.
• Final Rinse: The system is rinsed to remove chemical residues and prepared for production.
These automated steps ensure reliable and repeatable hygienic cleaning.
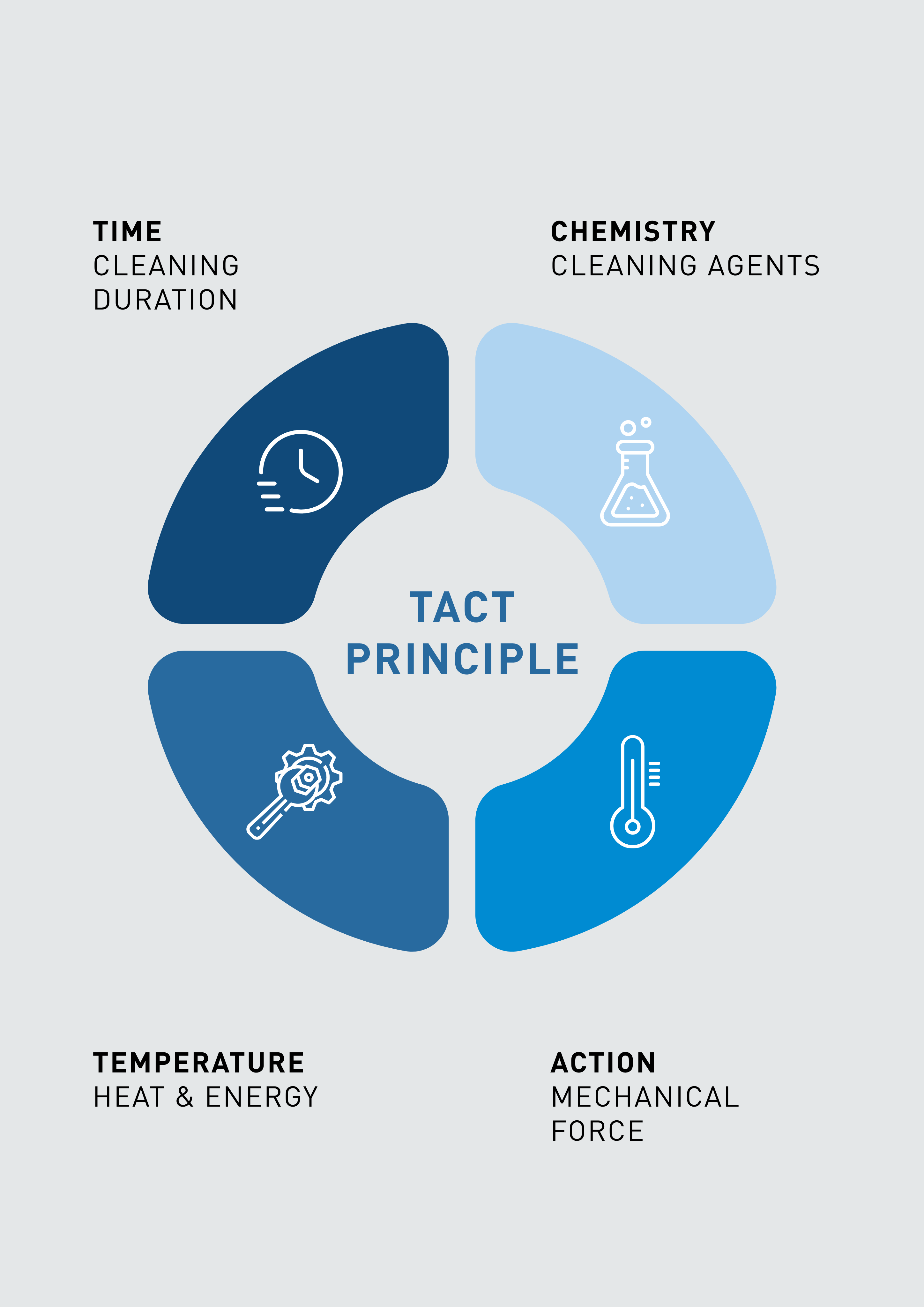
TACT Principle in CIP Cleaning
The cleaning performance of a CIP system depends on four key parameters known as the TACT principle.
• Time: Duration of the cleaning process
• Action: Mechanical force generated by fluid flow and turbulence
• Chemistry: Cleaning agents dissolve residues and deposits
• Temperature: Elevated temperatures improve cleaning efficiency
By balancing these four parameters, modern CIP cleaning systems achieve efficient cleaning while optimizing water, energy, and chemical consumption.
CIP Systems in Hygienic Industrial Production
CIP systems are used in hygienic production environments where reliable and repeatable cleaning of processing equipment is essential. They are applied across a wide range of industries that require strict hygiene standards and validated cleaning processes.
In food and beverage production, CIP systems remove residues such as fats, proteins, sugars, and starches from tanks, pipelines, heat exchangers, and processing units. Typical applications include dairy processing plants, beverage production lines, chocolate manufacturing, and sauce production systems.
In pharmaceutical manufacturing, CIP systems are used to clean reactors, fermenters, mixing vessels, and API production lines. Automated cleaning cycles support validated cleaning procedures and help manufacturers comply with regulatory standards such as GMP (Good Manufacturing Practice).
In cosmetics and personal care production, CIP systems clean equipment used for mixing tanks, emulsion reactors, and formulation systems for products such as creams, lotions, shampoos, and gels. Automated cleaning helps remove oils, emulsions, and formulation residues while minimizing manual cleaning effort and production downtime.
Advantages of CIP Systems
Modern CIP systems provide several important advantages for hygienic production environments
Key benefits include:
- Consistent and repeatable cleaning results
- Reduced production downtime
- Improved product safety and hygiene standards
- Lower manual cleaning effort
- Optimized water, energy, and chemical consumption
- Documented and validated cleaning cycles
These advantages make clean in place CIP systems an essential component of modern hygienic production processes.
CIP System vs. Manual Cleaning
Cleaning-in-Place (CIP) systems automate the cleaning of production equipment without requiring disassembly. Compared to manual cleaning, CIP ensures consistent hygiene, reduces downtime, and minimizes human error in industrial production environments.
| Aspect | CIP System | Manual Cleaning |
|---|---|---|
| Cleaning process | Automated cleaning cycles | Manual disassembly and cleaning |
| Production downtime | Minimal downtime | Longer production interruptions |
| Cleaning consistency | Repeatable and controlled | Dependent on manual work |
| Hygiene reliability | High and validated | Variable |
| Documentation | Automated monitoring | Limited documentation |
Challenges in Industrial Cleaning Processes
Maintaining hygienic production environments requires reliable cleaning processes that address a variety of technical and regulatory challenges. Production equipment often contains complex pipe systems, tanks, and processing units that must be cleaned thoroughly without interrupting operations.
Common challenges in industrial cleaning include:
• Removal of complex residues such as fats, proteins, starches, oils, or sticky product deposits
• Biofilm formation, where microorganisms attach to surfaces and become difficult to remove
• Cross contamination risks, particularly when different products are processed on the same production line
• Allergen removal, which is critical in food production to prevent unintended contamination
• Cleaning of complex equipment geometries, including pipelines, heat exchangers, and mixing systems
• Meeting validation and documentation requirements in regulated industries such as pharmaceuticals and food production
A properly designed CIP cleaning system helps manufacturers address these challenges by providing automated, controlled, and repeatable cleaning processes that ensure reliable hygiene standards.
-
ProCIP CompactModule CIP (solution d'entrée)

- Systèmes à usage unique et de récupération
- Capacités des réservoirs jusqu'à 20 000 l
- Débit jusqu'à 50m³/h
- Fonctions :
- préparation des solutions de nettoyage
- stockage des solutions de nettoyage (récupération)
- distribution de solutions de nettoyage
- suivi
- rapports
- Principales applications :
- nettoyage de réservoirs
- nettoyage des canalisations
- objets de nettoyage combinés
-
ProCIP SSystèmes CIP

- Systèmes à usage unique et de récupération
- Capacités des réservoirs jusqu'à 20 000 l
- Débit jusqu'à 50m³/h
- Fonctions :
- préparation des solutions de nettoyage
- stockage des solutions de nettoyage (récupération)
- distribution de solutions de nettoyage
- suivi
- rapports
- Principales applications :
- nettoyage de réservoirs
- nettoyage des canalisations
- objets de nettoyage combinés
-
ProCIP RecoveryUsine de récupération CIP

- Systèmes à usage unique et de récupération
- Capacités des réservoirs jusqu'à 20 000 l
- Débit jusqu'à 50m³/h
- Fonctions :
- préparation des solutions de nettoyage
- stockage des solutions de nettoyage (récupération)
- distribution de solutions de nettoyage
- suivi
- rapports
- Principales applications :
- nettoyage de réservoirs
- nettoyage des canalisations
- objets de nettoyage combinés
FAQs about CIP Systems
-
What does CIP mean in manufacturing?
CIP stands for Clean-in-Place and refers to automated cleaning systems used to clean production equipment internally without dismantling the machinery.
-
What is the purpose of a CIP system?
The purpose of a CIP system is to remove product residues, microorganisms, and contaminants from processing equipment to maintain hygienic production conditions.
-
Which industries use CIP systems?
CIP systems are widely used in the food, beverage, pharmaceutical, and cosmetics industries where strict hygiene standards are required.
-
How does a CIP cleaning system work?
A CIP cleaning system circulates water, cleaning chemicals, and sanitizing agents through processing equipment in controlled cleaning cycles to remove residues and contaminants.
-
What are the advantages of clean in place CIP systems?
Clean in place CIP systems reduce manual cleaning effort, minimize production downtime, improve hygiene consistency, and allow automated documentation of cleaning processes.

