-

CIP
Systems
食品加工、药品和化妆品的清洁解决方案
如果没有高效、可重复、可记录和可持续的清洁过程,食品加工,甚至化妆品或药品的生产就不可能保持高质量和高安全性。这不仅意味着要清除看得见的污垢,还意味着要消除看不见的污染,以实现安全卫生的生产。ProXES CIP 系统解决方案适用于食品加工、HPC 和制药加工。我们成功的清洁技术基于 TACT - 时间、作用、化学和温度的结合,以确保产品质量和设备的可用性。
What is a CIP System?
A CIP system is an automated cleaning technology used to clean production equipment internally without disassembling the machinery. Cleaning liquids such as water, detergents, and sanitizing solutions circulate through tanks, pipelines, and processing units to remove residues and contaminants.
Compared to manual cleaning methods, clean in place CIP systems provide controlled and repeatable cleaning cycles that improve hygiene and operational efficiency. Automated monitoring of cleaning parameters also allows cleaning processes to be documented and validated.
CIP systems are widely used in industries such as:
• Food and beverage processing
• Pharmaceutical manufacturing
• Cosmetics and personal care production
• Dairy and beverage industries
By automating the cleaning process, a CIP cleaning system helps manufacturers maintain reliable hygiene standards and efficient production workflows.
Typical CIP Process Steps
Industrial CIP systems follow a defined cleaning sequence designed to remove product residues, microorganisms, and contaminants from process equipment. These automated cleaning cycles allow tanks, pipelines, and processing units to be cleaned without dismantling the installation.
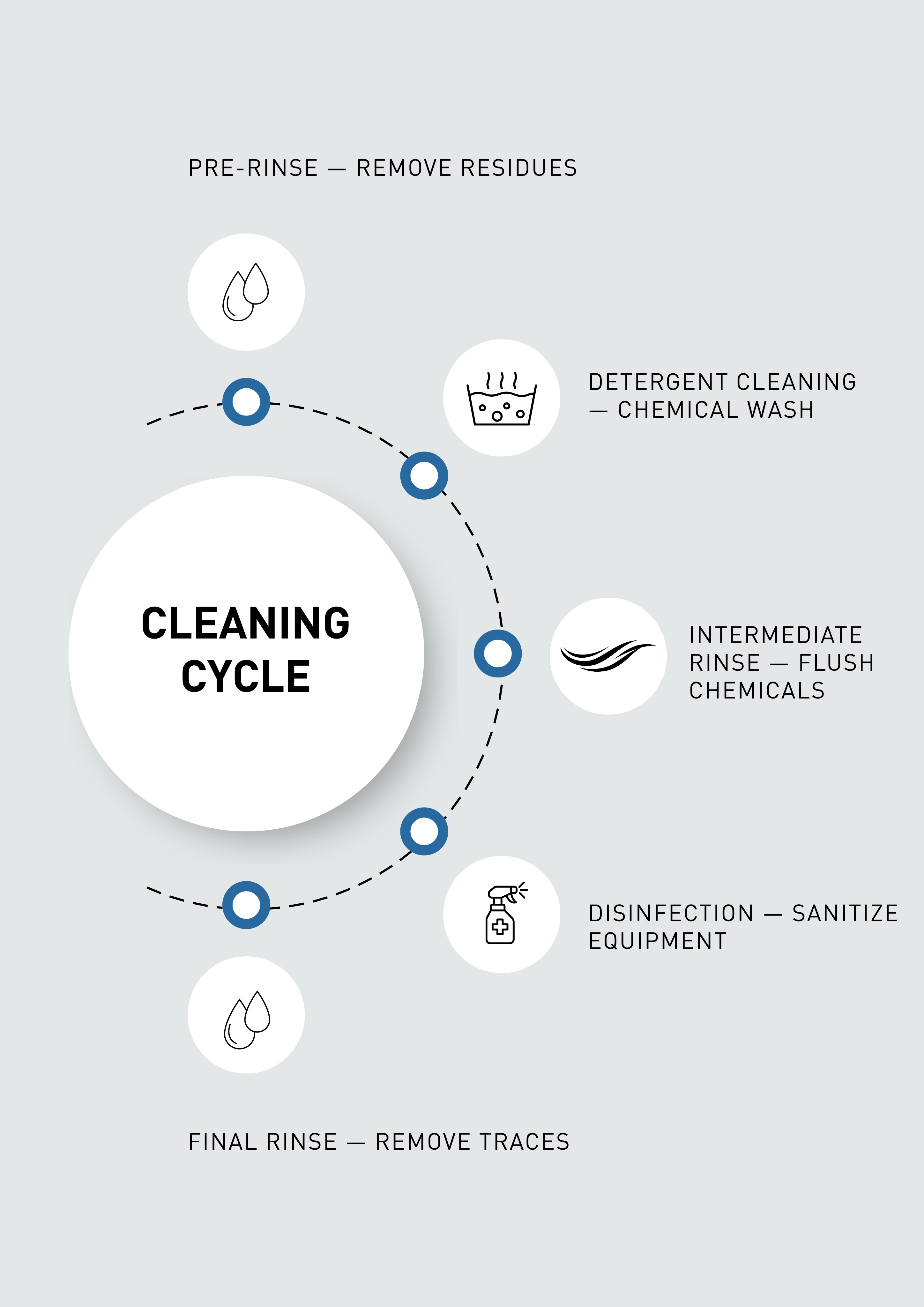
Cleaning Cycle Overview
A typical CIP cleaning cycle includes several stages:
• Pre-Rinse: Water removes loose product residues from the system.
• Detergent Cleaning: Alkaline or acidic cleaning agents dissolve fats, proteins, sugars, and other deposits.
• Intermediate Rinse: Remaining chemicals and dissolved residues are flushed out.
• Disinfection / Sanitization: Sanitizing agents eliminate microorganisms.
• Final Rinse: The system is rinsed to remove chemical residues and prepared for production.
These automated steps ensure reliable and repeatable hygienic cleaning.
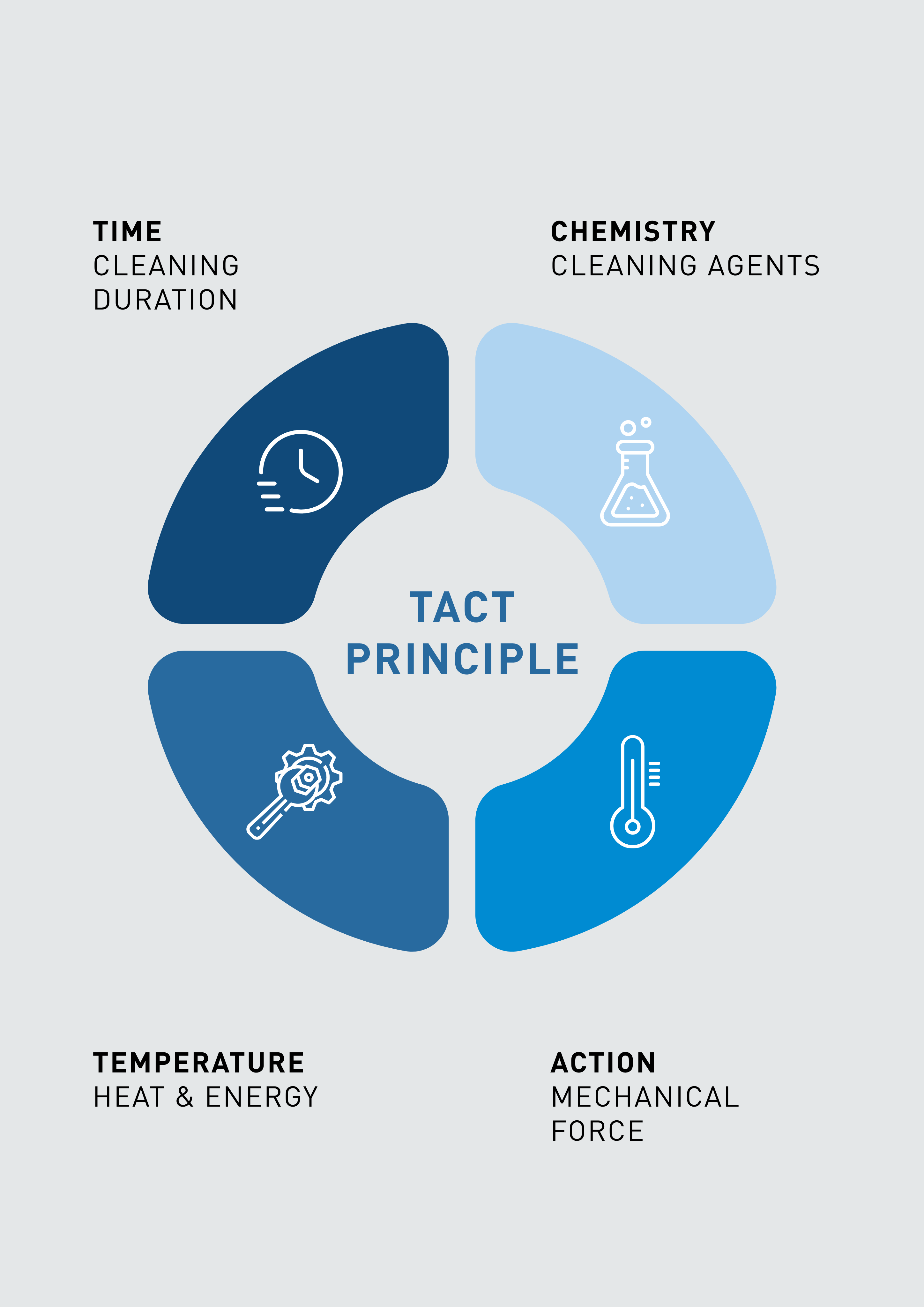
TACT Principle in CIP Cleaning
The cleaning performance of a CIP system depends on four key parameters known as the TACT principle.
• Time: Duration of the cleaning process
• Action: Mechanical force generated by fluid flow and turbulence
• Chemistry: Cleaning agents dissolve residues and deposits
• Temperature: Elevated temperatures improve cleaning efficiency
By balancing these four parameters, modern CIP cleaning systems achieve efficient cleaning while optimizing water, energy, and chemical consumption.
CIP Systems in Hygienic Industrial Production
CIP systems are used in hygienic production environments where reliable and repeatable cleaning of processing equipment is essential. They are applied across a wide range of industries that require strict hygiene standards and validated cleaning processes.
In food and beverage production, CIP systems remove residues such as fats, proteins, sugars, and starches from tanks, pipelines, heat exchangers, and processing units. Typical applications include dairy processing plants, beverage production lines, chocolate manufacturing, and sauce production systems.
In pharmaceutical manufacturing, CIP systems are used to clean reactors, fermenters, mixing vessels, and API production lines. Automated cleaning cycles support validated cleaning procedures and help manufacturers comply with regulatory standards such as GMP (Good Manufacturing Practice).
In cosmetics and personal care production, CIP systems clean equipment used for mixing tanks, emulsion reactors, and formulation systems for products such as creams, lotions, shampoos, and gels. Automated cleaning helps remove oils, emulsions, and formulation residues while minimizing manual cleaning effort and production downtime.
Advantages of CIP Systems
Modern CIP systems provide several important advantages for hygienic production environments
Key benefits include:
- Consistent and repeatable cleaning results
- Reduced production downtime
- Improved product safety and hygiene standards
- Lower manual cleaning effort
- Optimized water, energy, and chemical consumption
- Documented and validated cleaning cycles
These advantages make clean in place CIP systems an essential component of modern hygienic production processes.
CIP System vs. Manual Cleaning
Cleaning-in-Place (CIP) systems automate the cleaning of production equipment without requiring disassembly. Compared to manual cleaning, CIP ensures consistent hygiene, reduces downtime, and minimizes human error in industrial production environments.
| Aspect | CIP System | Manual Cleaning |
|---|---|---|
| Cleaning process | Automated cleaning cycles | Manual disassembly and cleaning |
| Production downtime | Minimal downtime | Longer production interruptions |
| Cleaning consistency | Repeatable and controlled | Dependent on manual work |
| Hygiene reliability | High and validated | Variable |
| Documentation | Automated monitoring | Limited documentation |
Challenges in Industrial Cleaning Processes
Maintaining hygienic production environments requires reliable cleaning processes that address a variety of technical and regulatory challenges. Production equipment often contains complex pipe systems, tanks, and processing units that must be cleaned thoroughly without interrupting operations.
Common challenges in industrial cleaning include:
• Removal of complex residues such as fats, proteins, starches, oils, or sticky product deposits
• Biofilm formation, where microorganisms attach to surfaces and become difficult to remove
• Cross contamination risks, particularly when different products are processed on the same production line
• Allergen removal, which is critical in food production to prevent unintended contamination
• Cleaning of complex equipment geometries, including pipelines, heat exchangers, and mixing systems
• Meeting validation and documentation requirements in regulated industries such as pharmaceuticals and food production
A properly designed CIP cleaning system helps manufacturers address these challenges by providing automated, controlled, and repeatable cleaning processes that ensure reliable hygiene standards.
-
ProCIP Compact一次性内部CIP清洗系统

- 一次性使用和回收系统
- 油箱容量高达20.000升
- 吞吐量高达50m³/h
- 功能:
- 清洁溶液的制备
- 清洁溶液的储存(回收)
- 清洁溶液的分配
- 监测
- 报告
- 主要应用:
- 罐体清洁
- 管道清洁
- 组合式的清洁对象
-
ProCIP S外接在线清洗批次单元

- 一次性使用和回收系统
- 存储罐容量高达20.000升
- 吞吐量高达50m³/h
- 功能:
- 清洁溶液的制备
- 清洁溶液的储存(回收)
- 清洁溶液的分配
- 监测
- 报告
- 主要应用:
- 罐体清洁
- 管道清洁
- 组合的清洁对象
**示例 -
ProCIP Recovery带回收重复利用的外部CIP系统

- 一次性使用和回收系统
- 油箱容量高达20.000升
- 吞吐量高达50m³/h
- 功能:
- 清洁溶液的制备
- 清洁溶液的储存(回收)
- 清洁溶液的分配
- 监测
- 报告
- 主要应用:
- 罐体清洁
- 管道清洁
- 组合式的清洁对象
FAQs about CIP Systems
-
What does CIP mean in manufacturing?
CIP stands for Clean-in-Place and refers to automated cleaning systems used to clean production equipment internally without dismantling the machinery.
-
What is the purpose of a CIP system?
The purpose of a CIP system is to remove product residues, microorganisms, and contaminants from processing equipment to maintain hygienic production conditions.
-
Which industries use CIP systems?
CIP systems are widely used in the food, beverage, pharmaceutical, and cosmetics industries where strict hygiene standards are required.
-
How does a CIP cleaning system work?
A CIP cleaning system circulates water, cleaning chemicals, and sanitizing agents through processing equipment in controlled cleaning cycles to remove residues and contaminants.
-
What are the advantages of clean in place CIP systems?
Clean in place CIP systems reduce manual cleaning effort, minimize production downtime, improve hygiene consistency, and allow automated documentation of cleaning processes.

